Received: Sat 04, Oct 2025
Accepted: Mon 27, Oct 2025
Abstract
Total nasal reconstruction is one of the most challenging reconstructive procedures in head and neck surgery, especially in irradiated tissues. We present the first case series of total nasal reconstruction using a fully 3D-printed integrated biomaterial through a staged surgery in three patients with a history of nasal cavity cancer treated by radiotherapy. Through the learning curve of these first patients, the surgical technique has been refined. In the final technique, the first stage of the procedure involved implanting the bioprosthesis in the patient’s forearm, under a skin flap corresponding to the angiosome of the radial artery. Subsequent surgeries under local anesthesia then involved touch-ups to better define the esthetic subunits of the nose, as well as to permeabilize and calibrate the nasal cavities. In a second surgery under general anesthesia, the radial forearm free flap with colonized implant was harvested and transferred to the face. The superficial temporal vessels served as recipient vessels. Esthetic results were satisfactory for all three patients. Although total nasal reconstruction is a challenging procedure with limited surgical options, this novel alternative can be a valuable addition to the reconstructive surgeon’s armamentarium.
Keywords
Total nasal reconstruction, bioprosthesis, biomaterial, radial forearm free flap, nasal tumor, total rhinectomy
1. Introduction
Total nasal reconstruction (TNR) is one of the most challenging reconstructive procedures in head and neck surgery, to such an extent that epithetic nasal reconstruction is generally considered the standard rehabilitation method [1]. However, epithetic nasal rehabilitation may be insufficient, impossible to achieve [2], or rejected by the patient. TNR often entails multiple surgical procedures, and requires a three-layer reconstruction: inner lining, nasal framework, and outer skin of the nose [3]. This is achieved via free flaps, costal or iliac bone grafts, covered with local flaps (e.g., forehead flaps) [4, 5]. All of these procedures carry a significant donor site morbidity and risk of failure [6]. Furthermore, the cosmetic outcomes can be disappointing, even in the hands of experts [7, 8].
When TNR is performed for oncological reasons, local flaps harvested from the face often transfer irradiated tissues, making them inadequate or unavailable for the repair of major nasal defects, and supporting the use of free flaps harvested from unirradiated areas [9]. Alloplastic material are a valid option since autologous grafts are more prone to complication in irradiated tissues [10]. In head and neck oncology, biomaterials are seen as a promising way to improve reconstruction outcomes and limit treatment morbidity [11, 12]. We herein describe the first cases of TNR in irradiated tissue with a fully 3D-printed integrated biomaterial via a staged surgery.
2. Methods
2.1. Planning and Bioprosthesis Design
The patients’ CT-scans were uploaded by the surgeon on Cerhum’s secured web-based platform (Link) used for communication and planning. The DICOM files were used to segment the patients’ bones and soft tissues as a base for future design discussions. In one case, the nose was already distorted by the disease in the initial CT-scan, so the patients’ epithesis was also scanned and segmented. The shape of the implant was designed to match the anatomical structure of a nose. Fixation holes were added to create channels for future attachment of the implant to soft tissues i.e., one in the dorsum, one in the columella, and one on each nasal ala. Then, three-dimensional gyroid porosities were implemented in the final volume using Autodesk Netfabb software, resulting in an implant with a porosity of approximately 64.5±5.2% [13, 14]. Pore size was designed with a diameter of 700 to 900 microns to allow sufficient space for vascularization and optimal hydrophilicity [13, 14].
2.2. Slurry Preparation (3D-Printing Material)
The photopolymerizable slurry was prepared by mixing hydroxyapatite powder with a photopolymerizable resin mixture. The resin was obtained by combining three types of additives: dispersants, photoinitiators, and acrylic monomers. The mixture was then processed in a three-roll mill to reduce the size of the agglomerates and to better homogenize the slurry. The resulting solid loading obtained was ≈50% [15].
2.3. 3D-Printing
The implant was produced using a V6000 stereolithography (VAT Photopolymerisation) machine by Prodways (Figure 1A). During the process, the slurry was spread on the working area in thin layers of 50 μm. After spreading each layer, a 345 nm UV light beam scanned the paste surface according to the CAD file slices in order to photopolymerize the layer of the part. Thus, the 3D shape of the implant was created layer-by-layer by photocuring of the paste material. The implants were then subjected to a high-temperature thermal cycle to remove the resin and densify the ceramic [16]. Finally, implant conformity was checked using a GOM scanner 3D-Atos Core 135. The resulting material is composed of 100% non-resorbable hydroxyapatite according to X-RAY diffractometry following the ISO 13779-3 : 2018 standard and presents microporosity and surface roughness that promotes cellular adhesion [14].
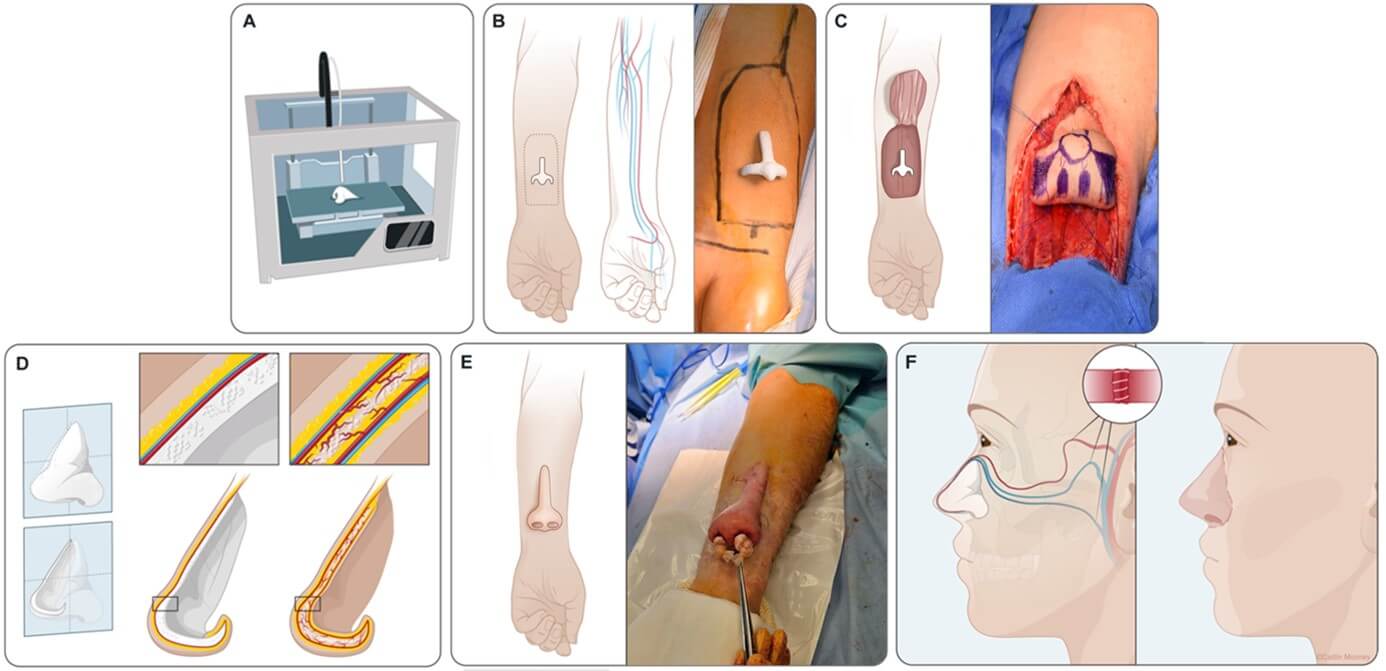
2.4. Surgery (Final Surgical Technique)
The surgical technique was gradually refined with the learning curve of the three first patients. We herein describe the final surgical technique, applied to the patient A, and recommended for its better outcomes. The patients gave their informed consent for the surgeries and the publication of this manuscript. TNR was planned into two procedures under general anesthesia and in the meantime some touch-up surgeries on the forearm, under local anesthesia.
During the first stage, the biomaterial was fostered on the forearm, which served as a bioreactor (Figures 1B & 1C). Then, 2.0 Ethilon® nylon sutures are passed through the fixation holes of the bioprosthesis (one in the dorsum, one in the columella, and one on each nasal ala), through the skin paddle, and tied in order to locate these structures during the next surgery under general anesthesia. The biomaterial is then entirely wrapped within the flap, without modeling it. A partial-thickness skin graft was used to cover the donor site. Implant colonization was controlled clinically, i.e., absence of biomaterial exposure. The second stage involved surgeries under local or no anesthesia, exclusively on the forearm (Figure 1D). The nostrils were perforated to make them permeable. Subsequent surgeries were performed to calibrate the nasal cavities and ensure that the biomaterial was not exposed and properly colonized by granulation tissue. Incisions and sutures were performed to refine the esthetic subunits of the nose, such as the nasal alae and dorsum.
During the third and final stage, the bioprosthesis was transferred to the face under general anesthesia. A time interval of at least two months after the implantation surgery was necessary to ensure complete biointegration. The radial forearm free flap was harvested, alongside the embedded bioprosthesis. Then, donor vessels were anastomosed to the temporal vessels and external jugular vein. The pedicle was tunneled in the infraorbital region. The flap was subsequently sutured to the recipient site using a two-layer interrupted suture (3.0 Vicryl® and 5.0 Prolene®) (Figure 1E). Small holes were drilled through the residual nasal bones, the zygomatic arch, and the anterior nasal spine. Then, the anchor stitches of 2.0 Ethilon, inserted through fixation holes of the biomaterial during the first stage, were passed through these perforated bones and tied. During this step, it is important to avoid compressing the pedicle. Nostril conformers must be left in place during the postoperative period.
3. Results - Case Reports
The final surgical technique, which has been refined through the learning curve of our patients, is described in Figure 1.
3.1. Patient A (Final Surgical Technique)
The last patient who underwent this procedure was a 60-year-old man at the time of the first surgery. He had been diagnosed with squamous cell carcinoma of the nasal cavity and had undergone total rhinectomy and adjuvant radiotherapy in 2021. A glued epithesis had been attempted but was ineffective. The biomaterial was placed in the radial forearm flap in November 2023 (Figure 2), but the surgery was complicated by deep venous thrombosis of the lower limb, revealing a prostatic cancer and requiring therapeutic anticoagulation. This delayed the subsequent surgeries to after discontinuation of anticoagulant drugs. Several touch-ups were performed on the forearm in the meantime, under local anesthesia: dorsum refinement and nostril calibration in July 2024, nostril permeabilization and calibration in December 2024, then nostril and nasal alae shaping with conformers in February 2025. The flap was transferred to the face in March 2025.
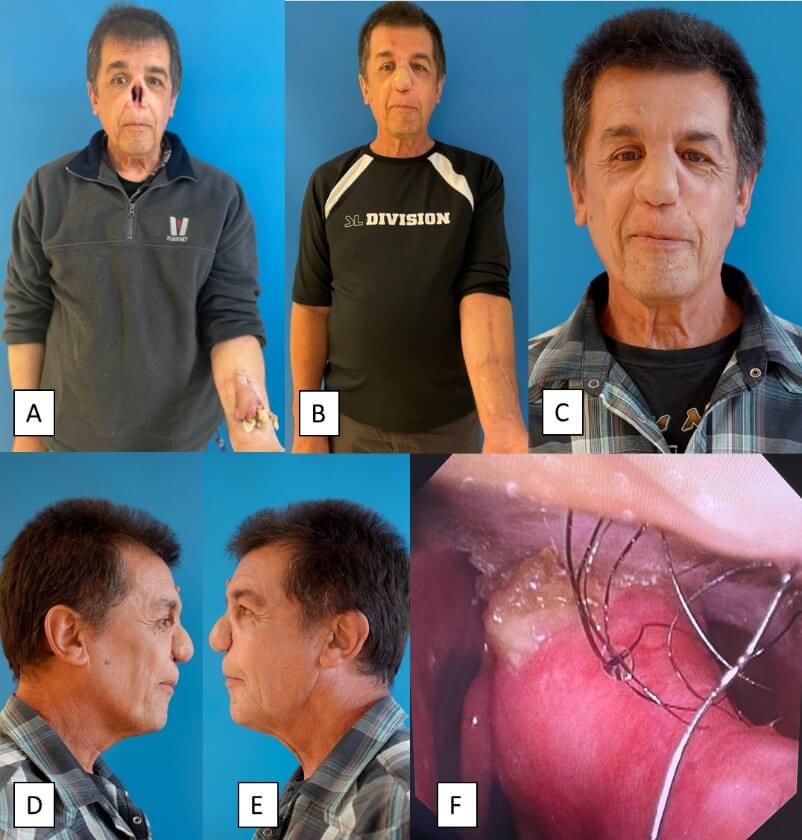
A) View of the nasal defect and bioprosthesis fostered on the patient’s forearm. B) Front view of the patient five months after the implantation surgery. C-E) Front and lateral views of the patient six months after the implantation surgery. F) Endonasal view showing the residual septum and both nasal cavities that are permeable. The right middle turbinate is seen on the left side of the picture.
Regarding quality-of-life assessments, his EuroQol Group–5 dimensions visual analogue scale (EQ-5D VAS) score was 70 at baseline and 70 at 3 months. The lack of improvement in this assessment is related, according to the patient, to prostate cancer and thrombophlebitis that occurred between the first and last assessments. The patient’s satisfaction with the cosmetic outcome of the nasal reconstruction was 10/10 on a Likert scale (1: poorly satisfied, 10: very satisfied). We applied an AI-based biological age estimation (FaceAge) on the face photographs before and after reconstruction [17], and the patient’s FaceAge before reconstruction (with epithesis in 2023 was 53 years (FaceAge minus Chronological Age deviation of 58 years) and the patient’s FaceAge after reconstruction in 2024 was 52.5 years (FaceAge minus chronological age deviation of 61 years) (Figure 2).
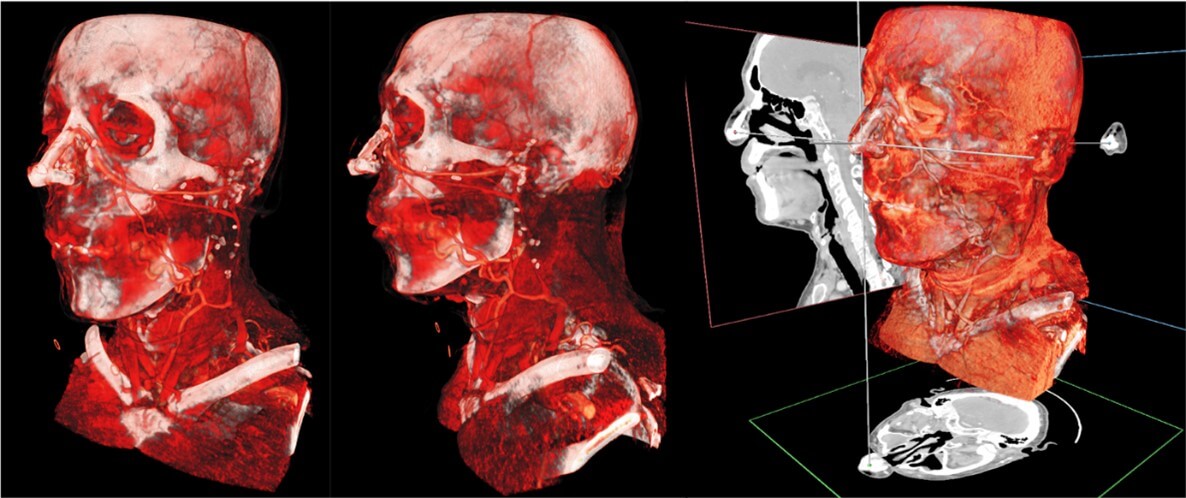
Figure 2 presents a comparison between baseline and 6 months post-surgery results for patient A. In June 2025, 3D models (Avatar Medical Vision) obtained from CT scan images allowed us to assess the good integration and position of the biomaterial (Figure 3).
3.2. Patient B
A 68-year-old man was diagnosed with squamous cell carcinoma on the nasal tip. He underwent partial nasal amputation and adjuvant radiotherapy in 2020. Moreover, nodal recurrence was treated with neck dissection and re-irradiation in 2021. Rehabilitation with a glued nasal epithesis was attempted but did not succeed.
In July 2023, this patient underwent biomaterial placement in the radial forearm flap. The bioprosthesis had a thinner design (like in patient A). The shape of the flap was trifoliate in order to mold the biomaterial (including nostrils and columella) with the radial skin paddle during the first procedure, at the time of implantation in the forearm, instead of simply wrapping it around the biomaterial. The surgery was complicated by a venous ischemia of the flap, resulting in a partial necrosis of the area intended for columella reconstruction. Two months later, the flap was transferred on the face and a costal graft was added for columella reconstruction. In December 2023, the flap underwent a touch-up (upper suspension) to correct the fall of the nasal tip and a costal graft was used for columella reconstruction. Another upper suspension was performed in April 2024 for the same reason. In March 2025, this patient had an EQ-5D VAS score of 90 and an EORTC QLQ-HN43 score of 54 vs. 55 at baseline in May 2023. A CT scan was performed in January 2025 to assess the integration of the biomaterial and provided satisfactory results, comparable to that of other patients. His satisfaction with the cosmetic outcomes on a Likert scale was 9/10. To this day, the patient wears nostril conformers only at night to maintain their caliber. Figure 4 shows a comparison between baseline and 20 months after surgery for patient B and an endonasal view.
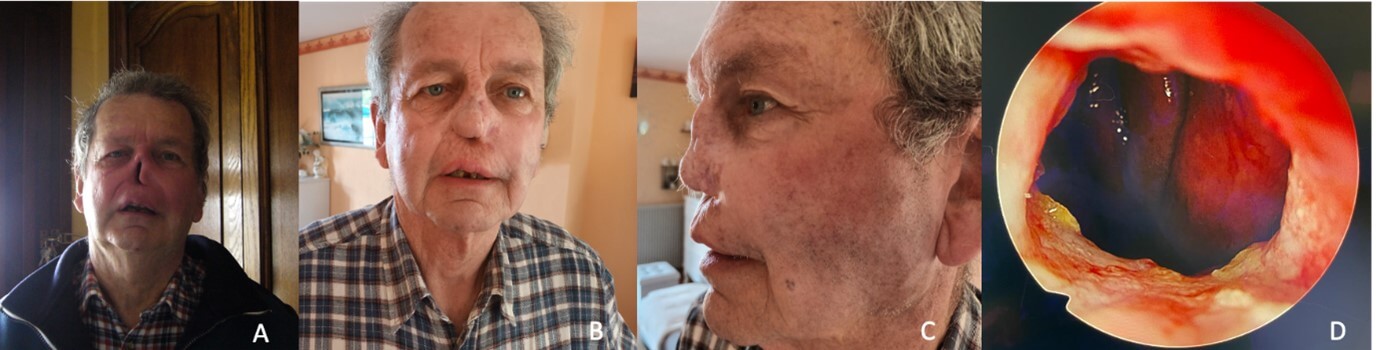
A) View of the nose before reconstruction. B & C) View of the nose 2 years after total nasal reconstruction (June 2025) by radial forearm free flap with embedded 3D-printed bioprosthesis in September 2023. D) Endonasal view.
3.3. Patient C
A 40-year-old woman presented to our clinic in 2012, after being misdiagnosed as a chronic atrophic polychondritis due to her history of progressive collapse of the nasal pyramid. She presented with a progressive necrosis of the anterior maxilla and oronasal communication. She was finally diagnosed with squamous cell carcinoma of the nasal cavity, leading to destruction of the nasal pyramid. She underwent definitive radiotherapy in 2012. Subsequently, her midfacial structures continued to melt and collapse, including the nasal cartilages, nasal bones, and piriform aperture. Rehabilitation with a nasal epithesis was attempted but failed due to lack of bone support in the superior maxilla. Therefore, she underwent several reconstructive surgeries, including a scapula free flap. However, all osseous and cartilaginous reconstructions “collapsed” over time.
The biomaterial was placed in the radial forearm flap in July 2022. Unlike in patients A and B, the bioprosthesis had a thicker design. As in patient B, we attempted to mold the biomaterial (including nostrils and columella) during the first procedure, with the trifoliate radial skin paddle, instead of simply wrapping it around the biomaterial. The suture lines of the flap were located on the nostrils and the columella, and the tension on these suture lines as well as the bulk of the implant were finally detrimental to the postoperative healing. Two months later, the flap was transferred to the nose. One month after that, a touch-up was performed on a 5 mm exposed part of the biomaterial at the level of each nasal ala, as a result of peripheral skin paddle retraction. Eight weeks after implantation, magnetic resonance imaging (MRI) and CT scan showed satisfactory biomaterial integration and vascularization without exposure to the skin or in the nasal cavity. The flap survived with no infection. In February 2023, the flap was refined at the level of the nasal dorsum, with a concurrent touch-up of a small exposed part of the biomaterial at the level of a nasal ala (on the area where the biomaterial was not covered by skin). Surgical revision was necessary to trim the biomaterial by 5mm using a Piezotome® and to cover it with the surrounding skin. The patient received an 8-week course of prophylactic antibiotherapy, and no clinical infection of the biomaterial was observed.
In December 2023, the biomaterial was removed because ptosis occurred due to its excessive size and lack of attachment to the residual bone, which was of very poor quality. The biomaterial was cut into pieces to be removed, and at that time we observed that several large vessels had formed in and around the biomaterial (Figure 5).
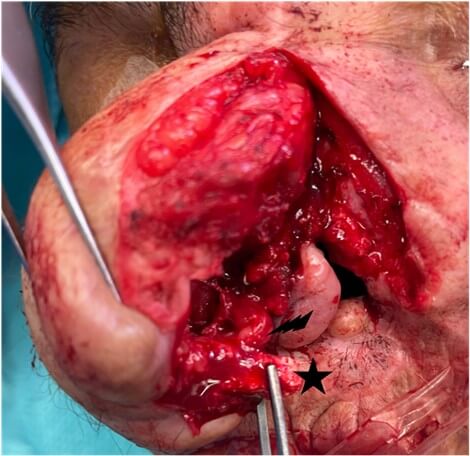
Black Star: Residual Biomaterial Reconstructing the Columella; Black Lightening: Blood Vessel.
It was replaced with a smaller biomaterial while keeping the free flap. This surgery was complicated by partial flap necrosis and biomaterial exposure at the columella. In August 2024, the exposure was covered by another radial forearm free flap, without prior fostering in the forearm. In April 2025, a new touch-up was required to remove a small exposed part of the biomaterial from the tip of the nose. Her EORTC QLQ-HN43 score improved from 54 to 46 at 34 months after surgery. This difference can be considered significant according to the literature [18, 19]. Her self-reported quality of life, as evaluated by the EQ-5D VAS, improved sharply between baseline (40/100) and 34 months after surgery (70/100), even though it was not the recommended surgical technique (suboptimal results). Her satisfaction with the cosmetic outcomes on a Likert scale was 7/10. She reported an improvement in her sense of smell, likely due to a more physiological nasal airflow. She started going out again after years of isolation.
Table 1 summarizes the demographic, patient history, complications and follow-up outcomes of the three patients.
Table. 1. Summary of the main outcomes for the three first patients undergoing
total nasal reconstruction with custom-made biomaterial and radial forearm free
flap.
|
Patient |
Demographics |
Medical history |
Date of implantation of the biomaterial |
Complications |
|
A |
60-year-old man |
Total rhinectomy and adjuvant radiotherapy |
November 2023 |
Transfer to the face delayed due to
metachronous cancer |
|
B |
68-year-old man |
Subtotal rhinectomy and adjuvant
radiotherapy |
July 2023 |
Partial necrosis of the flap (venous
ischemia) – biomaterial trimmed |
|
C |
40-year-old woman |
Nasal destruction by cancer and definitive
radiotherapy |
July 2022 |
Excessive bulk of the biomaterial causing
ptosis and exposure – biomaterial replaced with better design |
4. Discussion
In this case series, we reported the results and insights from the three first patients who underwent TNR in irradiated tissue with this novel technique, combining a hydroxyapatite porous biomaterial fostered in a radial forearm free flap. Although epithesis commonly offers better cosmetic results, this option failed in these three patients. The overall results of these three TNR were encouraging, demonstrating satisfactory cosmetic outcomes and proof of the integration and vascularization of the free flaps. The outcomes and the technique are gradually being refined and improved, which makes this approach promising given the ongoing advances in biomaterial sciences and reconstructive surgery.
To date, one case of TNR with a porous titanium plate has been described in 2018, without further development of this technique [20]. Moreover, in another article, Henry et al. used a titanium mesh and reported minor extrusion in two out of eight nasal reconstruction cases, although this rate was lower than the rates reported in the literature [21]. Indeed, titanium is prone to extrusion in irradiated tissue, as demonstrated by the cessation of its use in mandibular reconstruction [22, 23]. Another important point highlighted by our case series is the importance of transferring the bioprosthesis to the face only after the free flap has been properly shaped and molded, particularly at the level of the nasal alae. At the beginning of our learning curve, these touch-ups were done after the transfer on the patient’s face, as in for patient C, where the esthetic result was less satisfactory. Of note, the flap being insensate, all touch-ups can be performed with limited or without anesthesia.
For TNR, the biomaterial must be precisely tailored to the shape and size of the defect to repair. This has been made possible thanks to the advent of 3D printing [24]. The biomaterial must also have the ability to promote tissue integration, which can be achieved by adjusting the surface physicochemical properties of the biomaterial by designing a scaffold, adding stem cells, or using a bioreactor [11]. Moreover, the biomaterial must also be inert to limit the risk of immunological reaction [25]. Inspired by the living world (biomimicry), the biomaterial described in this article was made of hydroxyapatite, a bioceramic with a composition similar to that of natural bone [26]. The technique was chosen by the authors to promote the integration of the inert biomaterial and develop an intrinsic framework in the form of a scaffold, accelerating colonization by the surrounding tissues. In the cases described here, radiotherapy had induced hypovascularization of facial soft tissues [27, 28]. To increase the chances of biomaterial colonization, the patients’ arms were used as a bioreactor for a minimum period of 2 months. Two months after transplantation of the bioprosthesis to the face, clinical examination (absence of pain, local inflammation or biomaterial exposure) as well as imaging techniques conventionally used in the field of biomaterial (cone-beam combined to MRI) were both in favor of a complete integration of the bioprosthesis. Only small pieces of biomaterial were exposed due to skin flap retraction, but even these fragments showed blood vessel colonization. With a complete host integration of bioprosthesis after 12 to 36 months in our three patients, the risk period for biomaterial loss was presumably over. This suggests that the integration will most likely be long-lasting or definitive.
In clinical situations where anaplastology and reconstructive surgery have reached a stalemate, this additional option can significantly improve the patients’ cosmetic prognosis, and, consequently, their quality of life, level of activity, mood, and social integration [29, 30]. After several years without a nose and failed reconstructive surgeries or epithesis, patients showed an improvement in their quality of life.
We must emphasize that patient counseling is of the utmost importance in reconstructive planning for major nasal defects as the esthetic goals are likely to differ from one patient to another [31]. Patients must clearly be aware that even the best esthetic outcomes will be different from their native nose. In our study, patients had to keep the biomaterial embedded in their forearm during the fostering step. Although this can be perceived as esthetically displeasing, the forearm can be more easily concealed under clothing than in the case of paramedian forehead flaps, for example [31]. This vision can even bring hope to some patients. Moreover, 3D surface imaging of nasal structures before and after reconstruction can help with patient counseling [32], in addition to improving precision and outcomes [33].
Our approach, comprising only two general anesthesias, enabled TNR following the failure of multiple locoregional and free flaps by providing solid support for the nasal pyramid, with satisfactory esthetic outcomes. We evaluated patient satisfaction with the cosmetic outcomes on a scale from 1 to 10, as previously reported [3]. Their subjective assessment ranged from 9 to 10 for patients A and B, reflecting a general satisfaction with the procedure. Of note, this diversity regarding assessment tools underscores a lack of standardization. Overall, encouraging conclusions regarding esthetic results of TNR were also reported in a previous review [3]. This approach is relatively inexpensive and accessible, does not require a major expertise in TNR, and is associated with a limited donor site morbidity. Thanks to the advent of biomaterials, this approach offers new prospects, with limited morbidity, even in irradiated tissue.
From a cost-effectiveness point of view, the limited number of surgical procedures likely decreases the healthcare expenses. The average price of this biomaterial was about 6000€, though this cost may decrease in case of generalization of this technique. Cerhum, the company selling this custom-made biomaterial, is willing and able to scale up its production.
Among the limitations of this technique are the color mismatch and the complex reconstruction of the finest esthetic subunits of the nose, especially the shape of the nasal alae and columella. The radial forearm free flap may be too thick to adequately replace the skin of the nose. However, it remains one of the thinnest and most pliable free flaps. The use of successfully tested foundation makeup, though not used by our patient, is a simple solution to the color mismatch.
5. Conclusion
This is the first description of successful TNR through a staged procedure using a customized 3D-printed integrated hydroxyapatite biomaterial. While TNR is a challenging procedure with limited surgical options, this novel surgical alternative can be of great benefit to the reconstructive surgeon’s armamentarium. After substantial follow-up periods without late complications, this technique appears to be reliable, and merits further use in other patients.
Data Availability
The authors declare that the data associated with the publication of this manuscript is available upon reasonable request.
Funding
None.
Acknowledgments
We are indebted to the patient for his collaboration as well as to the staff of the head and neck surgery department for the surgeries and patient’s care. We would like to thank Raymond H. Maj, Carl Fridolin, and Andrew Warrington for evaluating the quality of the patient's reconstruction using Face Age AI.
REFERENCES
[1] Victoria
D'heygere, Stefan Mattheis, Kerstin Stähr, et al. “Epithetic nasal
reconstruction after total rhinectomy: Oncologic outcomes, immediate and
long-term adverse effects, and quality of life.” J Plast Reconstr Aesthet
Surg, vol. 74, no. 3, pp. 625-631, 2021. View at: Publisher Site | PubMed
[2] Vincent
Vander Poorten, Jeroen Meulemans, Pierre Delaere “Midface prosthetic
rehabilitation.” Curr Opin Otolaryngol Head Neck Surg, vol. 24, no. 2,
pp. 98-109, 2016. View at: Publisher Site | PubMed
[3] Andrea
Migliorelli, Rossella Sgarzani, Giovanni Cammaroto, et al. “Reconstructive
Options after Oncological Rhinectomy: State of the Art.” Healthcare (Basel),
vol. 11, no. 12, pp. 1785, 2023. View at: Publisher Site | PubMed
[4] David A Shaye
“The history of nasal reconstruction.” Curr Opin Otolaryngol Head Neck Surg,
vol. 29, no. 4, pp. 259-264, 2021. View at: Publisher Site | PubMed
[5] V Darsonval,
D Arnaud, J-B Duron, et al. “Full thickness reconstruction of the nasal
pyramid.” Ann Chir Plast Esthet, vol. 58, no. 5, pp. 544-600, 2013. View
at: Publisher Site | PubMed
[6] Florin-Vlad
Hodea, Cristian-Sorin Hariga, Eliza-Maria Bordeanu-Diaconescu, et al.
“Assessing Donor Site Morbidity and Impact on Quality of Life in Free Flap
Microsurgery: An Overview.” Life (Basel), vol. 15, no. 1, pp. 36, 2024.
View at: Publisher
Site | PubMed
[7] Jithin John,
Rohun Gupta, Anne Grossbauer, et al. “Outcomes Associated with Nasal
Reconstruction Post-Rhinectomy: A Narrative Review.” Arch Plast Surg,
vol. 49, no. 2, pp. 184-194, 2022. View at: Publisher Site | PubMed
[8] Young Ji
Park, Gyu Hyeon Kwon, Jun Oh Kim, et al. “Reconstruction of nasal ala and tip
following skin cancer resection.” Arch Craniofac Surg, vol. 20, no. 6,
pp. 382-387, 2019. View at: Publisher Site | PubMed
[9] Konstantinos
Gasteratos, Georgia-Alexandra Spyropoulou, Kongkrit Chaiyasate “Microvascular
Reconstruction of Complex Nasal Defects: Case Reports and Review of the
Literature.” Plast Reconstr Surg Glob Open, vol. 8, no. 7, pp. e3003,
2020. View at: Publisher Site | PubMed
[10] M A
ElKhashab, I A W Radi, A H Elkhadem “Implant prognosis in irradiated versus
non-irradiated nasal, orbital and auricular sites.” Int J Oral Maxillofac
Surg, vol. 49, no. 5, pp. 636-648, 2020. View at: Publisher Site | PubMed
[11] Lorenzo
Moroni, Jennifer H. Elisseeff “Biomaterials engineered for integration.” Materials
Today, vol. 11, no. 5, pp. 44-51, 2008. View at: Publisher Site
[12] Tim J
Phillips “Total nasal reconstruction: a review of the past and present, with a
peak into the future.” Curr Opin Otolaryngol Head Neck Surg, vol. 27,
no. 5, pp. 420-425, 2019. View at: Publisher Site | PubMed
[13] Islam
Bouakaz, Ehsan Sadeghian Dehkord, Sylvain Meille, et al. “3D printed triply
periodic minimal surfaces calcium phosphate bone substitute: The effect of
porosity design on mechanical properties.” Ceramics International, vol.
50, no. 2, Part A, pp. 2623-2636, 2024. View at: Publisher Site
[14] Islam
Bouakaz, Christophe Drouet, David Grossin, et al. “Hydroxyapatite 3D-printed
scaffolds with Gyroid-Triply periodic minimal surface porous structure:
Fabrication and an in vivo pilot study in sheep.” Acta Biomater, vol.
170, pp. 580-595, 2023. View at: Publisher Site | PubMed
[15] Laurent Le
Guéhennec, Dorien Van Hede, Erwan Plougonven, et al. “In vitro and in vivo
biocompatibility of calcium-phosphate scaffolds three-dimensional printed by
stereolithography for bone regeneration.” J Biomed Mater Res A, vol.
108, no. 3, pp. 412-425, 2020. View at: Publisher
Site | PubMed
[16] Goffard R,
Sforza T, Clarinval A, et al. “Additive manufacturing of biocompatible
ceramics.” Advances in Production Engineering & Management, vol. 8,
no. 2, pp. 96-106, 2013. View at: Publisher Site
[17] Dennis
Bontempi, Osbert Zalay, Danielle S Bitterman, et al. “FaceAge, a deep learning
system to estimate biological age from face photographs to improve
prognostication: a model development and validation study.” Lancet Digit
Health, vol. 7, no. 6, pp. 100870, 2025. View at: Publisher Site | PubMed
[18] Susanne
Singer, Eva Hammerlid, Iwona M Tomaszewska, et al. “Methodological approach for
determining the Minimal Important Difference and Minimal Important Change
scores for the European Organisation for Research and Treatment of Cancer Head
and Neck Cancer Module (EORTC QLQ-HN43) exemplified by the Swallowing scale.” Qual
Life Res, vol. 31, no. 3, pp. 841-853, 2022. View at: Publisher Site | PubMed
[19] D Osoba, G
Rodrigues, J Myles, et al. “Interpreting the significance of changes in
health-related quality-of-life scores.” J Clin Oncol, vol. 16, no. 1,
pp. 139-144, 1998. View at: Publisher Site | PubMed
[20] Quentin
Qassemyar, Nathaniel Assouly, Yoni Madar, et al. “Total nasal reconstruction
with 3D custom made porous titanium prosthesis and free thoracodorsal artery
perforator flap: A case report.” Microsurgery, vol. 38, no. 5, pp.
567-571, 2018. View at: Publisher Site | PubMed
[21] Eric L Henry,
Robert D Hart, S Mark Taylor, et al. “Total nasal reconstruction: use of a
radial forearm free flap, titanium mesh, and a paramedian forehead flap.” J
Otolaryngol Head Neck Surg, vol. 39, no. 6, pp. 697-702, 2010. View at: Publisher Site | PubMed
[22] Fabienne
Haroun, Nadia Benmoussa, François Bidault, et al. “Outcomes of mandibular
reconstruction using three-dimensional custom-made porous titanium prostheses.”
J Stomatol Oral Maxillofac Surg, vol. 124, no. 1S, pp. 101281, 2023.
View at: Publisher Site | PubMed
[23] J K Ryu, R L
Stern, M G Robinson, et al. “Mandibular reconstruction using a titanium plate:
the impact of radiation therapy on plate preservation.” Int J Radiat Oncol
Biol Phys, vol. 32, no. 3, pp. 627-634, 1995. View at: Publisher Site | PubMed
[24] Neerajha
Nagarajan, Agnes Dupret-Bories, Erdem Karabulut, et al. “Enabling personalized
implant and controllable biosystem development through 3D printing.” Biotechnol
Adv, vol. 36, no. 2, pp. 521-533, 2018. View at: Publisher Site | PubMed
[25] Matthew J
Webbe “Embracing Simplicity in Biomaterials Design.” Biomater Biosyst,
vol. 6, pp. 100043, 2022. View at: Publisher Site | PubMed
[26] Didarul
Bhuiyan, John Middleton, Rina Tannenbaum “Development of
hydroxyapatite-mediated synthesis of collagen-based copolymers for application
as bio scaffolds in bone regeneration.” MRS Online Proceedings Library,
vol. 1621, no. 1, pp. 53-58, 2014. View at: Publisher
Site
[27] Emely
Kjellsson Lindblom, Susanta Hui, Jamison Brooks, et al. “Radiation-induced
Vascular Damage and the Impact on the Treatment Outcome of Stereotactic Body
Radiotherapy.” Anticancer Res, vol. 39, no. 6, pp. 2721-2727, 2019. View
at: Publisher Site | PubMed
[28] Peter
Grabham, Preety Sharma “The effects of radiation on angiogenesis.” Vasc Cell,
vol. 5, no. 1, pp. 19, 2013. View at: Publisher Site | PubMed
[29] C Becker, A M
Becker, K K K Dahlem, et al. “Aesthetic and functional outcomes in patients
with a nasal prosthesis.” Int J Oral Maxillofac Surg, vol. 46, no. 11,
pp. 1446-1450, 2017. View at: Publisher Site | PubMed
[30] Sanne E
Moolenburgh 1, Marc A M Mureau, Sarah L Versnel, et al. “The impact of nasal
reconstruction following tumour resection on psychosocial functioning, a
clinical-empirical exploration.” Psychooncology, vol. 18, no. 7, pp.
747-752, 2009. View at: Publisher Site | PubMed
[31] Heather K
Schopper, Shekhar K Gadkaree, Jessyka G Lighthall “Approach to Major Nasal
Reconstruction: Benefits of Staged Surgery and Use of Technology.” Facial
Plast Surg Clin North Am, vol. 32, no. 2, pp. 199-210, 2024. View at: Publisher Site | PubMed
[32] Glynnis De
Greve, Ronit Malka, Erica Barnett, et al. “Three-Dimensional Technology in
Rhinoplasty.” Facial Plast Surg, vol. 38, no. 5, pp. 483-487, 2022. View at: Publisher Site | PubMed
[33] Raisa Chowdhury, Nisreen Al-Musaileem, Karanvir S Raman, et al. “3D Printing in Nasal Reconstruction: Application-Based Evidence on What Works, When, and Why.” Biomedicines, vol. 13, no. 6, pp. 1434, 2025. View at: Publisher Site | PubMed
